
Another limitation of Dalton’s theory is that it does not account for allotropes such as diamond and graphite, both of which have completely different properties although they both contain only carbon.(3) On the other hand, atoms of different elements have different properties and differ in mass and. (2) A particular element’s atoms have the same mass, size, and other properties. This disproves Dalton’s postulate that atoms of different elements combine in simple, whole-number ratios to form compounds. The postulates of Daltons Atomic Theory are listed below: (1) Atoms are small, indivisible particles that make up all matter. In certain complex organic compounds such as sugar (C 11 H 22 O 11 ), the individual elements do not combine in a simple whole number ratio.For example, calcium and argon are isobars with each having an atomic mass of 40 amu. However, this does not account for isobars, which refers to different elements that share the same mass number. According to Dalton’s theory, atoms of different elements are different in all respects. 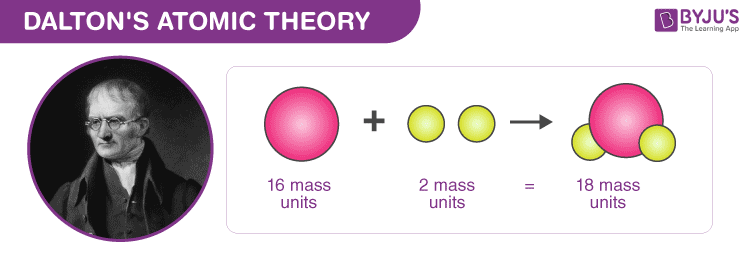
Chlorine is one such atom that has two isotopes with mass numbers 35 and 37. However, further studies indicate that atoms of some elements vary in their density and mass. Dalton’s theory states that atoms of the same element are identical to each other.This disproves Dalton’s assumption that atoms are indivisible. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
